A Survey of Transcriptome Complexity in Sus Scrofa Using Single-molecule Long-read Sequencing
Third-generation sequencing (also known every bit long-read sequencing) is a grade of DNA sequencing methods currently under active development.[one]
Third generation sequencing technologies have the capability to produce substantially longer reads than second generation sequencing.[1] Such an reward has critical implications for both genome science and the study of biological science in full general. Nonetheless, third generation sequencing data accept much higher fault rates than previous technologies, which can complicate downstream genome assembly and assay of the resulting data.[2] These technologies are undergoing active evolution and it is expected that there will be improvements to the high error rates. For applications that are more tolerant to mistake rates, such as structural variant calling, third generation sequencing has been found to outperform existing methods, even at a low depth of sequencing coverage.[iii]
Electric current technologies [edit]
Sequencing technologies with a unlike approach than second-generation platforms were start described as "tertiary-generation" in 2008–2009.[4]
At that place are several companies currently at the heart of third generation sequencing engineering science development, namely, Pacific Biosciences, Oxford Nanopore Technology, Quantapore (CA-U.s.a.), and Stratos (WA-U.s.). These companies are taking fundamentally unlike approaches to sequencing single Deoxyribonucleic acid molecules.
PacBio developed the sequencing platform of single molecule real fourth dimension sequencing (SMRT), based on the properties of zippo-style waveguides. Signals are in the form of fluorescent light emission from each nucleotide incorporated by a Dna polymerase bound to the bottom of the zL well.
Oxford Nanopore's engineering science involves passing a Dna molecule through a nanoscale pore structure and then measuring changes in electrical field surrounding the pore; while Quantapore has a different proprietary nanopore approach. Stratos Genomics spaces out the Dna bases with polymeric inserts, "Xpandomers", to circumvent the point to noise claiming of nanopore ssDNA reading.
Too notable is Helicos's unmarried molecule fluorescence approach, only the company entered bankruptcy in the fall of 2015.
Advantages [edit]
Longer reads [edit]
In comparing to the electric current generation of sequencing technologies, third generation sequencing has the obvious advantage of producing much longer reads. Information technology is expected that these longer read lengths will alleviate numerous computational challenges surrounding genome associates, transcript reconstruction, and metagenomics amidst other important areas of modernistic biology and medicine.[1]
It is well known that eukaryotic genomes including primates and humans are complex and have large numbers of long repeated regions. Short reads from second generation sequencing must resort to approximative strategies in order to infer sequences over long ranges for assembly and genetic variant calling. Pair end reads have been leveraged by 2d generation sequencing to combat these limitations. Still, exact fragment lengths of pair ends are often unknown and must also exist approximated likewise. Past making long reads lengths possible, tertiary generation sequencing technologies have clear advantages.
Epigenetics [edit]
Epigenetic markers are stable and potentially heritable modifications to the Dna molecule that are not in its sequence. An example is DNA methylation at CpG sites, which has been found to influence gene expression. Histone modifications are another instance. The electric current generation of sequencing technologies rely on laboratory technique such equally Scrap-sequencing for the detection of epigenetic markers. These techniques involve tagging the Deoxyribonucleic acid strand, breaking and filtering fragments that contain markers, followed past sequencing. Third generation sequencing may enable direct detection of these markers due to their distinctive betoken from the other four nucleotide bases.[5]
Portability and speed [edit]

MinION Portable Cistron Sequencer, Oxford Nanopore Technologies
Other important advantages of third generation sequencing technologies include portability and sequencing speed.[6] Since minimal sample preprocessing is required in comparing to 2d generation sequencing, smaller equipments could be designed. Oxford Nanopore Technology has recently commercialized the MinION sequencer. This sequencing machine is roughly the size of a regular USB flash bulldoze and can exist used readily past connecting to a laptop. In add-on, since the sequencing process is not parallelized across regions of the genome, information could exist collected and analyzed in real time. These advantages of third generation sequencing may be well-suited in hospital settings where quick and on-site data collection and analysis is demanded.
Challenges [edit]
| | Parts of this article (those related to long-read sequencing technologies producing low-accuracy reads. While truthful 5 years ago, circular consensus reads with the PacBio Sequel Ii long-read sequencer tin easily achieve an even college read accurateness than hybrid genome assembly with a combination of other sequencers. [1] PMID 31885515, 28364362, 31406327, 31897449, 31483244) need to exist updated. (January 2020) |
Third generation sequencing, as it currently stands, faces important challenges mainly surrounding accurate identification of nucleotide bases; error rates are still much higher compared to second generation sequencing.[ii] This is generally due to instability of the molecular machinery involved. For example, in PacBio's single molecular and existent time sequencing applied science, the Deoxyribonucleic acid polymerase molecule becomes increasingly damaged as the sequencing process occurs.[2] Additionally, since the process happens chop-chop, the signals given off by private bases may be blurred by signals from neighbouring bases. This poses a new computational challenge for deciphering the signals and consequently inferring the sequence. Methods such as Subconscious Markov Models, for instance, accept been leveraged for this purpose with some success.[five]
On average, different individuals of the human population share nigh 99.nine% of their genes. In other words, approximately merely 1 out of every thousand bases would differ between any 2 person. The loftier error rates involved with third generation sequencing are inevitably problematic for the purpose of characterizing private differences that exist between members of the same species.
Genome assembly [edit]
Genome assembly is the reconstruction of whole genome DNA sequences. This is by and large washed with 2 fundamentally unlike approaches.
Reference alignment [edit]
When a reference genome is available, every bit 1 is in the case of human, newly sequenced reads could simply be aligned to the reference genome in order to narrate its properties. Such reference based assembly is quick and easy but has the disadvantage of "hiding" novel sequences and large re-create number variants. In addition, reference genomes do not yet be for nigh organisms.
De novo associates [edit]
De novo assembly is the alternative genome assembly approach to reference alignment. It refers to the reconstruction of whole genome sequences entirely from raw sequence reads. This method would exist chosen when there is no reference genome, when the species of the given organism is unknown as in metagenomics, or when there exist genetic variants of interest that may non exist detected past reference genome alignment.
Given the brusk reads produced by the current generation of sequencing technologies, de novo assembly is a major computational trouble. It is unremarkably approached past an iterative procedure of finding and connecting sequence reads with sensible overlaps. Various computational and statistical techniques, such as de bruijn graphs and overlap layout consensus graphs, have been leveraged to solve this problem. Nonetheless, due to the highly repetitive nature of eukaryotic genomes, accurate and complete reconstruction of genome sequences in de novo assembly remains challenging. Pair end reads take been posed as a possible solution, though verbal fragment lengths are often unknown and must exist approximated.[vii]
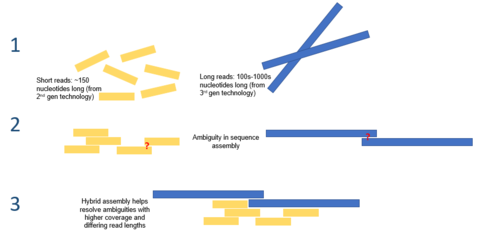
Hybrid associates – the utilize of reads from 3rd gen sequencing platforms with shorts reads from 2nd gen platforms – may exist used to resolve ambiguities that be in genomes previously assembled using 2nd generation sequencing. Short second generation reads have likewise been used to correct errors that exist in the long 3rd generation reads.
Hybrid associates [edit]
Long read lengths offered by tertiary generation sequencing may alleviate many of the challenges currently faced by de novo genome assemblies. For instance, if an unabridged repetitive region can exist sequenced unambiguously in a single read, no computation inference would be required. Computational methods have been proposed to alleviate the outcome of loftier fault rates. For example, in 1 study, it was demonstrated that de novo assembly of a microbial genome using PacBio sequencing alone performed superior to that of second generation sequencing.[8]
Third generation sequencing may also be used in conjunction with 2d generation sequencing. This arroyo is often referred to equally hybrid sequencing. For example, long reads from third generation sequencing may be used to resolve ambiguities that exist in genomes previously assembled using 2d generation sequencing. On the other hand, curt second generation reads have been used to correct errors in that exist in the long tertiary generation reads. In full general, this hybrid approach has been shown to improve de novo genome assemblies significantly.[9]
Epigenetic markers [edit]
DNA methylation (DNAm) – the covalent modification of DNA at CpG sites resulting in attached methyl groups – is the best understood component of epigenetic machinery. DNA modifications and resulting gene expression can vary across cell types, temporal development, with genetic ancestry, tin can alter due to environmental stimuli and are heritable. After the discovery of DNAm, researchers have too plant its correlation to diseases like cancer and autism.[10] In this disease etiology context DNAm is an important avenue of farther research.
Advantages [edit]
The current virtually mutual methods for examining methylation state require an assay that fragments Deoxyribonucleic acid before standard second generation sequencing on the Illumina platform. As a result of short read length, information regarding the longer patterns of methylation are lost.[v] 3rd generation sequencing technologies offer the capability for single molecule real-time sequencing of longer reads, and detection of DNA modification without the aforementioned assay.[11]
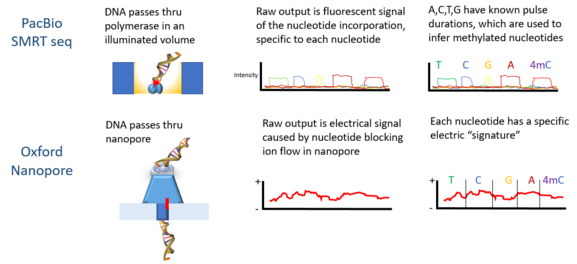
PacBio SMRT technology and Oxford Nanopore can use unaltered Deoxyribonucleic acid to detect methylation.
Oxford Nanopore Technologies' MinION has been used to detect DNAm. As each DNA strand passes through a pore, information technology produces electrical signals which have been found to exist sensitive to epigenetic changes in the nucleotides, and a subconscious Markov model (HMM) was used to analyze MinION data to detect 5-methylcytosine (5mC) DNA modification.[5] The model was trained using synthetically methylated E. coli Dna and the resulting signals measured by the nanopore technology. So the trained model was used to discover 5mC in MinION genomic reads from a human cell line which already had a reference methylome. The classifier has 82% accuracy in randomly sampled singleton sites, which increases to 95% when more stringent thresholds are practical.[5]
Other methods address dissimilar types of DNA modifications using the MinION platform. Stoiber et al. examined 4-methylcytosine (4mC) and half-dozen-methyladenine (6mA), along with 5mC, and likewise created software to directly visualize the raw MinION data in a human-friendly fashion.[12] Here they found that in E. coli, which has a known methylome, event windows of 5 base pairs long can be used to divide and statistically analyze the raw MinION electrical signals. A straightforward Mann-Whitney U test tin can find modified portions of the E. coli sequence, besides as further split the modifications into 4mC, 6mA or 5mC regions.[12]
It seems likely that in the time to come, MinION raw data will be used to detect many dissimilar epigenetic marks in Deoxyribonucleic acid.
PacBio sequencing has as well been used to observe DNA methylation. In this platform, the pulse width – the width of a fluorescent light pulse – corresponds to a specific base. In 2010 information technology was shown that the interpulse distance in control and methylated samples are different, and in that location is a "signature" pulse width for each methylation type.[11] In 2012 using the PacBio platform the bounden sites of Deoxyribonucleic acid methyltransferases were characterized.[xiii] The detection of N6-methylation in C Elegans was shown in 2015.[14] Dna methylation on N 6-adenine using the PacBio platform in mouse embryonic stalk cells was shown in 2016.[15]
Other forms of DNA modifications – from heavy metals, oxidation, or UV damage – are besides possible avenues of inquiry using Oxford Nanopore and PacBio third generation sequencing.
Drawbacks [edit]
Processing of the raw data – such as normalization to the median signal – was needed on MinION raw data, reducing real-time capability of the technology.[12] Consistency of the electrical signals is still an issue, making it hard to accurately call a nucleotide. MinION has low throughput; since multiple overlapping reads are hard to obtain, this farther leads to accurateness problems of downstream Deoxyribonucleic acid modification detection. Both the hidden Markov model and statistical methods used with MinION raw data crave repeated observations of Dna modifications for detection, meaning that individual modified nucleotides need to be consistently present in multiple copies of the genome, eastward.grand. in multiple cells or plasmids in the sample.
For the PacBio platform, also, depending on what methylation you expect to discover, coverage needs can vary. As of March 2017, other epigenetic factors like histone modifications accept not been discoverable using tertiary-generation technologies. Longer patterns of methylation are frequently lost considering smaller contigs still demand to be assembled.
Transcriptomics [edit]
Transcriptomics is the study of the transcriptome, unremarkably past characterizing the relative abundances of messenger RNA molecules the tissue under study. According to the primal dogma of molecular biology, genetic data flows from double stranded Dna molecules to single stranded mRNA molecules where they tin be readily translated into function protein molecules. By studying the transcriptome, one tin can gain valuable insight into the regulation of gene expressions.
While expression levels as the gene level can be more or less accurately depicted by 2d generation sequencing, transcript-level information even so remains an important claiming.[16] Equally a upshot, the role of alternative splicing in molecular biology remains largely elusive. Third generation sequencing technologies hold promising prospects in resolving this issue past enabling sequencing of mRNA molecules at their total lengths.
Alternative splicing [edit]
Alternative splicing (Equally) is the process by which a single gene may give ascension to multiple singled-out mRNA transcripts and consequently unlike poly peptide translations.[17] Some evidence suggests that Equally is a ubiquitous phenomenon and may play a central office in determining the phenotypes of organisms, peculiarly in complex eukaryotes; all eukaryotes contain genes consisting of introns that may undergo AS. In particular, it has been estimated that AS occurs in 95% of all homo multi-exon genes.[eighteen] AS has undeniable potential to influence myriad biological processes. Advancing knowledge in this expanse has critical implications for the study of biology in general.
Transcript reconstruction [edit]
The current generation of sequencing technologies produce only curt reads, putting tremendous limitation on the power to detect distinct transcripts; curt reads must exist opposite engineered into original transcripts that could accept given rise to the resulting read observations.[nineteen] This task is farther complicated by the highly variable expression levels across transcripts, and consequently variable read coverages across the sequence of the gene.[19] In improver, exons may be shared among individual transcripts, rendering unambiguous inferences essentially impossible.[17] Existing computational methods make inferences based on the accumulation of short reads at various sequence locations often by making simplifying assumptions.[xix] Cufflinks takes a parsimonious arroyo, seeking to explicate all the reads with the fewest possible number of transcripts.[xx] On the other hand, StringTie attempts to simultaneously estimate transcript abundances while assembling the reads.[19] These methods, while reasonable, may non always place real transcripts.
A report published in 2008 surveyed 25 unlike existing transcript reconstruction protocols.[16] Its show suggested that existing methods are generally weak in assembling transcripts, though the power to detect individual exons are relatively intact.[sixteen] According to the estimates, average sensitivity to detect exons across the 25 protocols is 80% for Caenorhabditis elegans genes.[sixteen] In comparison, transcript identification sensitivity decreases to 65%. For man, the study reported an exon detection sensitivity averaging to 69% and transcript detection sensitivity had an boilerplate of a mere 33%.[16] In other words, for human, existing methods are able to identify less than one-half of all existing transcript.
Third generation sequencing technologies have demonstrated promising prospects in solving the problem of transcript detection as well as mRNA abundance estimation at the level of transcripts. While mistake rates remain loftier, third generation sequencing technologies have the capability to produce much longer read lengths.[21] Pacific Bioscience has introduced the iso-seq platform, proposing to sequence mRNA molecules at their full lengths.[21] It is anticipated that Oxford Nanopore will put forth similar technologies. The trouble with higher mistake rates may be alleviated by supplementary high quality short reads. This approach has been previously tested and reported to reduce the error charge per unit by more than 3 folds.[22]
Metagenomics [edit]
Metagenomics is the analysis of genetic fabric recovered directly from ecology samples.
Advantages [edit]
The main advantage for third-generation sequencing technologies in metagenomics is their speed of sequencing in comparison to 2d generation techniques. Speed of sequencing is important for case in the clinical setting (i.due east. pathogen identification), to let for efficient diagnosis and timely clinical actions.
Oxford Nanopore's MinION was used in 2015 for real-time metagenomic detection of pathogens in complex, high-background clinical samples. The outset Ebola virus (EBV) read was sequenced 44 seconds afterward data conquering.[23] There was uniform mapping of reads to genome; at least one read mapped to >88% of the genome. The relatively long reads allowed for sequencing of a virtually-complete viral genome to loftier accuracy (97–99% identity) directly from a primary clinical sample.[23]
A common phylogenetic mark for microbial customs diverseness studies is the 16S ribosomal RNA factor. Both MinION and PacBio's SMRT platform take been used to sequence this factor.[24] [25] In this context the PacBio error charge per unit was comparable to that of shorter reads from 454 and Illumina's MiSeq sequencing platforms.[ citation needed ]
Drawbacks [edit]
MinION's high error rate (~10-40%) prevented identification of antimicrobial resistance markers, for which unmarried nucleotide resolution is necessary. For the same reason, eukaryotic pathogens were non identified.[23] Ease of carryover contamination when re-using the same flow jail cell (standard wash protocols don't work) is too a business concern. Unique barcodes may allow for more multiplexing. Furthermore, performing accurate species identification for bacteria, fungi and parasites is very difficult, as they share a larger portion of the genome, and some only differ by <5%.
The per base sequencing toll is still significantly more than that of MiSeq. Withal, the prospect of supplementing reference databases with total-length sequences from organisms below the limit of detection from the Sanger approach;[24] this could possibly greatly help the identification of organisms in metagenomics.
References [edit]
- ^ a b c Bleidorn, Christoph (2016-01-02). "Tertiary generation sequencing: engineering science and its potential affect on evolutionary biodiversity research". Systematics and Biodiversity. 14 (1): 1–eight. doi:10.1080/14772000.2015.1099575. ISSN 1477-2000. S2CID 85991118.
- ^ a b c Gupta, Pushpendra One thousand. (2008-11-01). "Single-molecule DNA sequencing technologies for hereafter genomics research". Trends in Biotechnology. 26 (xi): 602–611. doi:x.1016/j.tibtech.2008.07.003. PMID 18722683.
- ^ Tham, Cheng Yong (2020-03-03). "NanoVar: accurate characterization of patients' genomic structural variants using depression-depth nanopore sequencing". Genome Biology. 21 (Article number: 56): 56. doi:10.1186/s13059-020-01968-7. PMC7055087. PMID 32127024.
- ^ Check Hayden, Erika (2009-02-06). "Genome sequencing: the third generation". Nature News. 457 (7231): 768–769. doi:10.1038/news.2009.86. PMID 19212365.
- ^ a b c d due east Simpson, Jared T.; Workman, Rachael; Zuzarte, Philip C.; David, Matei; Dursi, Lewis Jonathan; Timp, Winston (2016-04-04). "Detecting DNA Methylation using the Oxford Nanopore Technologies MinION sequencer". bioRxiv10.1101/047142.
- ^ Schadt, East. E.; Turner, S.; Kasarskis, A. (2010-10-xv). "A window into tertiary-generation sequencing". Human Molecular Genetics. 19 (R2): R227–R240. doi:ten.1093/hmg/ddq416. ISSN 0964-6906. PMID 20858600.
- ^ Li, Ruiqiang; Zhu, Hongmei; Ruan, Jue; Qian, Wubin; Fang, Xiaodong; Shi, Zhongbin; Li, Yingrui; Li, Shengting; Shan, Gao (2010-02-01). "De novo associates of human genomes with massively parallel short read sequencing". Genome Inquiry. twenty (2): 265–272. doi:10.1101/gr.097261.109. ISSN 1088-9051. PMC2813482. PMID 20019144.
- ^ Mentum, Chen-Shan; Alexander, David H.; Marks, Patrick; Klammer, Aaron A.; Drake, James; Heiner, Cheryl; Clum, Alicia; Copeland, Alex; Huddleston, John (2013-06-01). "Nonhybrid, finished microbial genome assemblies from long-read SMRT sequencing data". Nature Methods. 10 (half-dozen): 563–569. doi:ten.1038/nmeth.2474. ISSN 1548-7091. PMID 23644548. S2CID 205421576.
- ^ Goodwin, Sara; Gurtowski, James; Ethe-Sayers, Scott; Deshpande, Panchajanya; Schatz, Michael C.; McCombie, Westward. Richard (2015-xi-01). "Oxford Nanopore sequencing, hybrid error correction, and de novo assembly of a eukaryotic genome". Genome Research. 25 (11): 1750–1756. doi:10.1101/gr.191395.115. ISSN 1088-9051. PMC4617970. PMID 26447147.
- ^ Fraser, Hunter B.; Lam, Lucia 50.; Neumann, Sarah M.; Kobor, Michael Southward. (2012-02-09). "Population-specificity of human Dna methylation". Genome Biological science. 13 (ii): R8. doi:10.1186/gb-2012-13-2-r8. ISSN 1474-760X. PMC3334571. PMID 22322129.
- ^ a b Flusberg, Benjamin A.; Webster, Dale R.; Lee, Jessica H.; Travers, Kevin J.; Olivares, Eric C.; Clark, Tyson A.; Korlach, Jonas; Turner, Stephen W. (2010-06-01). "Direct detection of Deoxyribonucleic acid methylation during unmarried-molecule, existent-time sequencing". Nature Methods. 7 (six): 461–465. doi:x.1038/nmeth.1459. PMC2879396. PMID 20453866.
- ^ a b c Stoiber, Marcus H.; Quick, Joshua; Egan, Rob; Lee, Ji Eun; Celniker, Susan Due east.; Neely, Robert; Loman, Nicholas; Pennacchio, Len; Brown, James B. (2016-12-fifteen). "De novo Identification of Deoxyribonucleic acid Modifications Enabled by Genome-Guided Nanopore Indicate Processing". bioRxiv10.1101/094672.
- ^ Clark, T. A.; Murray, I. A.; Morgan, R. D.; Kislyuk, A. O.; Spittle, K. Due east.; Boitano, M.; Fomenkov, A.; Roberts, R. J.; Korlach, J. (2012-02-01). "Characterization of DNA methyltransferase specificities using single-molecule, existent-time Deoxyribonucleic acid sequencing". Nucleic Acids Research. 40 (iv): e29. doi:10.1093/nar/gkr1146. ISSN 0305-1048. PMC3287169. PMID 22156058.
- ^ Greer, Eric Lieberman; Blanco, Mario Andres; Gu, Lei; Sendinc, Erdem; Liu, Jianzhao; Aristizábal-Corrales, David; Hsu, Chih-Hung; Aravind, L.; He, Chuan (2015). "DNA Methylation on N6-Adenine in C. elegans". Cell. 161 (iv): 868–878. doi:10.1016/j.cell.2015.04.005. PMC4427530. PMID 25936839.
- ^ Wu, Tao P.; Wang, Tao; Seetin, Matthew G.; Lai, Yongquan; Zhu, Shijia; Lin, Kaixuan; Liu, Yifei; Byrum, Stephanie D.; Mackintosh, Samuel K. (2016-04-21). "DNA methylation on N6-adenine in mammalian embryonic stalk cells". Nature. 532 (7599): 329–333. Bibcode:2016Natur.532..329W. doi:10.1038/nature17640. ISSN 0028-0836. PMC4977844. PMID 27027282.
- ^ a b c d e Steijger, Tamara; Abril, Josep F.; Engström, Pär G.; Kokocinski, Felix; The RGASP Consortium; Hubbard, Tim J.; Guigó, Roderic; Harrow, Jennifer; Bertone, Paul (2013-12-01). "Cess of transcript reconstruction methods for RNA-seq". Nature Methods. 10 (12): 1177–1184. doi:10.1038/nmeth.2714. ISSN 1548-7091. PMC3851240. PMID 24185837.
- ^ a b Graveley, Brenton R. (2001). "Culling splicing: increasing diversity in the proteomic globe". Trends in Genetics. 17 (2): 100–107. doi:10.1016/s0168-9525(00)02176-4. PMID 11173120.
- ^ Pan, Qun; Shai, Ofer; Lee, Leo J.; Frey, Brendan J.; Blencowe, Benjamin J. (2008-12-01). "Deep surveying of alternative splicing complexity in the human being transcriptome by high-throughput sequencing". Nature Genetics. twoscore (12): 1413–1415. doi:ten.1038/ng.259. ISSN 1061-4036. PMID 18978789. S2CID 9228930.
- ^ a b c d Pertea, Mihaela; Pertea, Geo M.; Antonescu, Corina M.; Chang, Tsung-Cheng; Mendell, Joshua T.; Salzberg, Steven 50. (2015-03-01). "StringTie enables improved reconstruction of a transcriptome from RNA-seq reads". Nature Biotechnology. 33 (3): 290–295. doi:10.1038/nbt.3122. ISSN 1087-0156. PMC4643835. PMID 25690850.
- ^ Trapnell, Cole; Williams, Brian A.; Pertea, Geo; Mortazavi, Ali; Kwan, Gordon; van Baren, Marijke J.; Salzberg, Steven L.; Wold, Barbara J.; Pachter, Lior (2010-05-01). "Transcript assembly and quantification past RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation". Nature Biotechnology. 28 (5): 511–515. doi:x.1038/nbt.1621. ISSN 1087-0156. PMC3146043. PMID 20436464.
- ^ a b Abdel-Ghany, Salah E.; Hamilton, Michael; Jacobi, Jennifer L.; Ngam, Peter; Devitt, Nicholas; Schilkey, Faye; Ben-Hur, Asa; Reddy, Anireddy S. N. (2016-06-24). "A survey of the sorghum transcriptome using single-molecule long reads". Nature Communications. 7: 11706. Bibcode:2016NatCo...711706A. doi:10.1038/ncomms11706. ISSN 2041-1723. PMC4931028. PMID 27339290.
- ^ Au, Kin Fai; Underwood, Jason G.; Lee, Lawrence; Wong, Wing Hung (2012-10-04). "Improving PacBio Long Read Accuracy by Curt Read Alignment". PLOS ONE. 7 (10): e46679. Bibcode:2012PLoSO...746679A. doi:ten.1371/journal.pone.0046679. ISSN 1932-6203. PMC3464235. PMID 23056399.
- ^ a b c Greninger, Alexander L.; Naccache, Samia North.; Federman, Scot; Yu, Guixia; Mbala, Placide; Bres, Vanessa; Stryke, Doug; Boutonniere, Jerome; Somasekar, Sneha (2015-01-01). "Rapid metagenomic identification of viral pathogens in clinical samples past real-time nanopore sequencing analysis". Genome Medicine. 7: 99. doi:10.1186/s13073-015-0220-ix. ISSN 1756-994X. PMC4587849. PMID 26416663.
- ^ a b Schloss, Patrick D.; Jenior, Matthew 50.; Koumpouras, Charles C.; Westcott, Sarah L.; Highlander, Sarah K. (2016-01-01). "Sequencing 16S rRNA gene fragments using the PacBio SMRT Dna sequencing organisation". PeerJ. four: e1869. doi:10.7717/peerj.1869. PMC4824876. PMID 27069806.
- ^ Benítez-Páez, Alfonso; Portune, Kevin J.; Sanz, Yolanda (2016-01-01). "Species-level resolution of 16S rRNA factor amplicons sequenced through the MinION™ portable nanopore sequencer". GigaScience. 5: 4. doi:10.1186/s13742-016-0111-z. ISSN 2047-217X. PMC4730766. PMID 26823973.
Source: https://en.wikipedia.org/wiki/Third-generation_sequencing
0 Response to "A Survey of Transcriptome Complexity in Sus Scrofa Using Single-molecule Long-read Sequencing"
Post a Comment